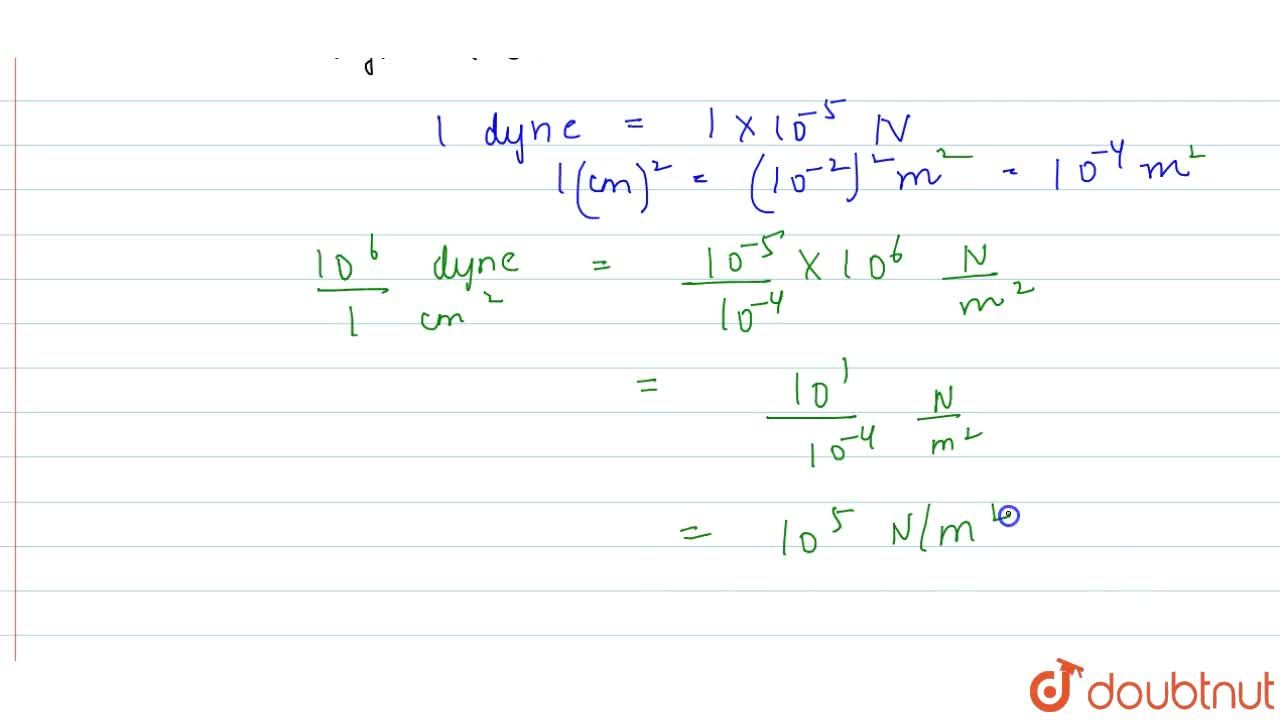convert a pressure of one atmospheric(=105 N m2)into dyne cm-2 could anyone please explain with detailed steps - Physics - Units And Measurements - 4719700 | Meritnation.com

A torque of 10^(8) dyne - cm is applied to a fly wheel of mass 10 kg and radius of gyration 50 cm. What is the resultant angular acceleration ?
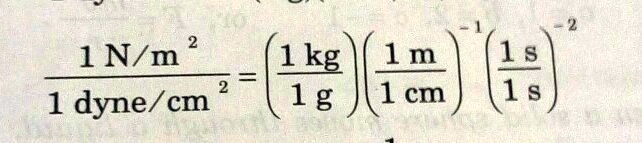
SOLVED: 'solve it plz conversion factor between newton and dyne 1 N/m 1 dyne/ cm 1kg 1n | (4 1 g cm 1 '

Convert the folowing (a) 5 dyne/cm^2 into N/m^2 (b) 10 g/cm^3 into Kg/m^3 - Physics - Units And Measurements - 12979011 | Meritnation.com